Taukah anda apa yang terjadi apabila suatu atom atau molekul menyerap (absorpsi) foton?
Sebelum menjawab pertanyaan di atas, kita harus mengingat terlebih dahulu tentang teori Niel Bohr. Menurut Bohr, tingkat energi elektronik pada atom dan molekul bersifat diskrit, artinya tidak semua tingkat energi ada, namun hanya tingkat-tingkat energi tertentu yang dimiliki oleh atom dan molekul. Misalnya pada atom hidrogen, tingkat energi utamanya yang mungkin adalah -13,6 eV/n2 dengan n adalah bilangan bulat (1, 2, 3, 4, dst). Atom hidrogen tidak mempunya energi utama dengan n=1/2 , 3/2 atau bilangan tidak bulat lainnya.
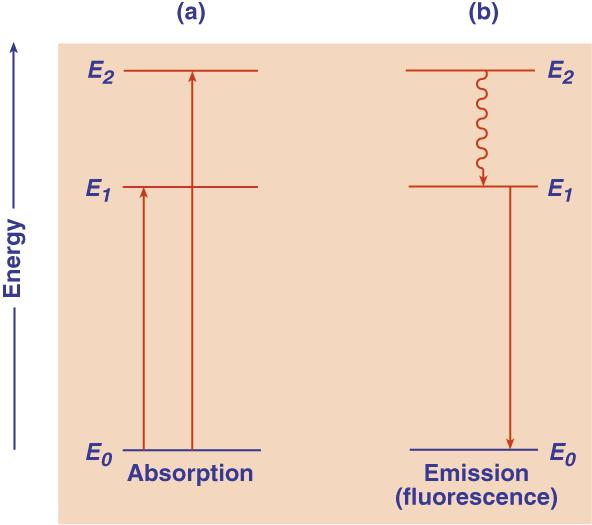
Fenomena absorpsi cahaya (foton) berkaitan dengan perpindahan elektron di antara tingkat-tingkat energi tersebut. Pada keadaan dasar, elektron akan menempati tingkat energi terendah yang disebut tingkat energi dasar (E0). Absorpsi foton akan menyebabkan atom atau molekul tereksitasi, dengan kata lain elektron pada atom atau molekul itu akan berpindah ke tingkat energi yang lebih tinggi (E1 atau E2).
Gambar 1. Tingkat energi dasar dan dua tingkat energi tereksitasi
(diambil dari http://www.life.illinois.edu/govindjee/biochem494/Abs.html)
Segera setelah elektron mengalami eksitasi, elektron tersebut akan kembali ke keadaan dasar dengan melepaskan radiasi dalam bentuk foton. Gejala ini disebut emisi. Apabila peristiwa emisi tidak disertai oleh perubahan spin elektron maka dinamakan flouresensi.
Gejala absorpsi dan flouresensi banyak digunakan dalam spektroskopi untuk identifikasi dan uji kuantitatif atom. Misalnya pada spektroskopi serapan atom (SSA) untuk uji kuantitatif logam di dalam sampel.
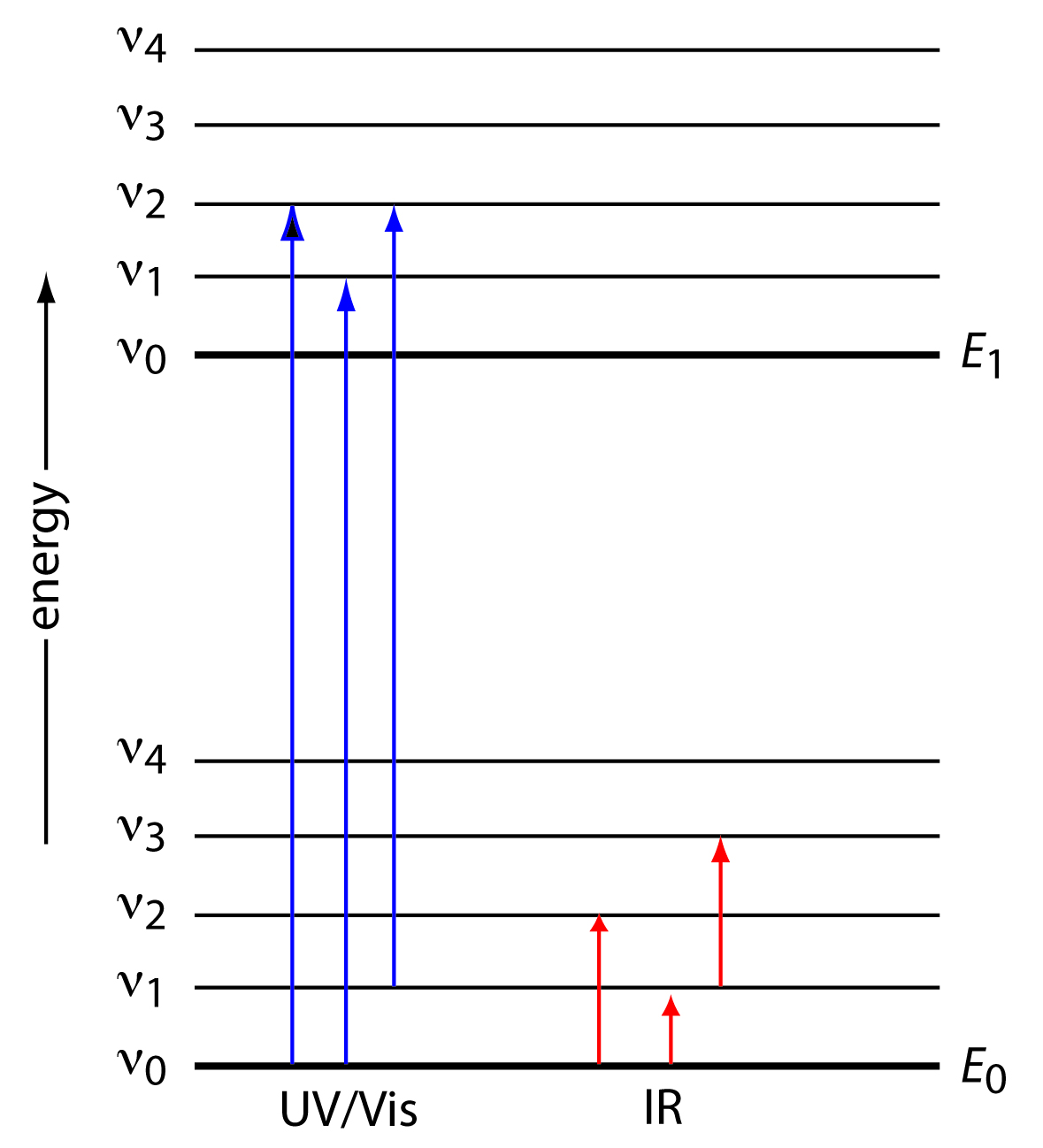
Pada molekul diatomik dan poliatomik, selain tingkat energi utama, juga terdapat tingkat energi vibrasional dan rotasional yang berkaitan dengan kemampuan molekul untuk bervibrasi dan berortasi. Perbedaan antar energi vibrasi dan rotasi ini sangat kecil apabila dibandingkan dengan perbedaan antar tingkat energi utama.
Gambar 2. Diagram yang menunjukkan dua tingkat energi elektronik utama (E0 dan E1) dengan masing-masing tingkat energi vibrasinya.
(diambil dari http://chemwiki.ucdavis.edu/@api/deki/files/12508/Figure10.15.jpg)
Pada gambar di atas terlihat pada eksitasi elektronik dari E0 dan E1 pada molekul lebih komplek daripada pada atom karena adanya banyak tingkat energi vibrasi dan rotasi pada kedua tingkat energi utama itu. Tingkat-tingkat energi rotasi dan vibrasi sangat berdekatan dan seolah menciptakan spektra yang bersifat kontinyu seperti pada gambar di bawah.
Gambar 3. Spektra absorpsi/emisi pada atom (a) dan molekul (b)
(diambil dari http://www.monzir-pal.net/Instrumental%20Analysis/Contents/Properties_of_electromag_rad.htm)
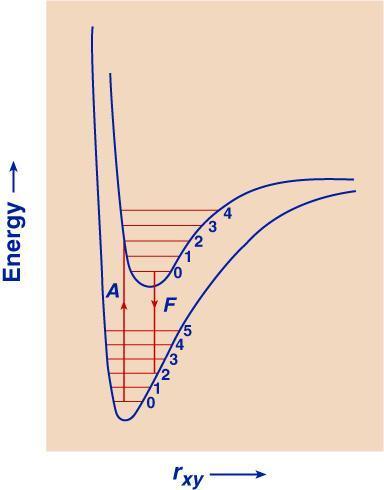
Gambar 2 merupakan ilustrasi sederhana tingkat-tingkat energi vibrasi dan rotasi pada molekul. Penggambaran yang lebih lengkap adalah Gambar 4 dan Gambar 5.
Gambar 4. Kurva energi potensial untuk keadaan dasar dan keadaan tereksitasi pada molekul
Bersambung............